
We use cookies to offer you the best experience on our site. You can find out more about the cookies we use or disable them in the Cookie settings
Single-use systems have become essential in modern biopharmaceutical manufacturing, enabling safer, more flexible, and more efficient production processes. Designed for one-time use and delivered ready-to-use, assembled in cleanroom and sterilized, these assemblies eliminate cleaning and cross-contamination risks while simplifying validation and reducing downtime.
For pharmaceutical and biotech manufacturers facing increasingly complex aseptic operations, custom single-use solutions provide the flexibility to adapt processes quickly, secure sterile fluid transfer, and accelerate time-to-market.
With more than 20 years of expertise in aseptic processing, clean injection molding, and ready-to-use assemblies, Aseptic Technologies delivers tailored single-use systems designed to optimize critical life science applications.
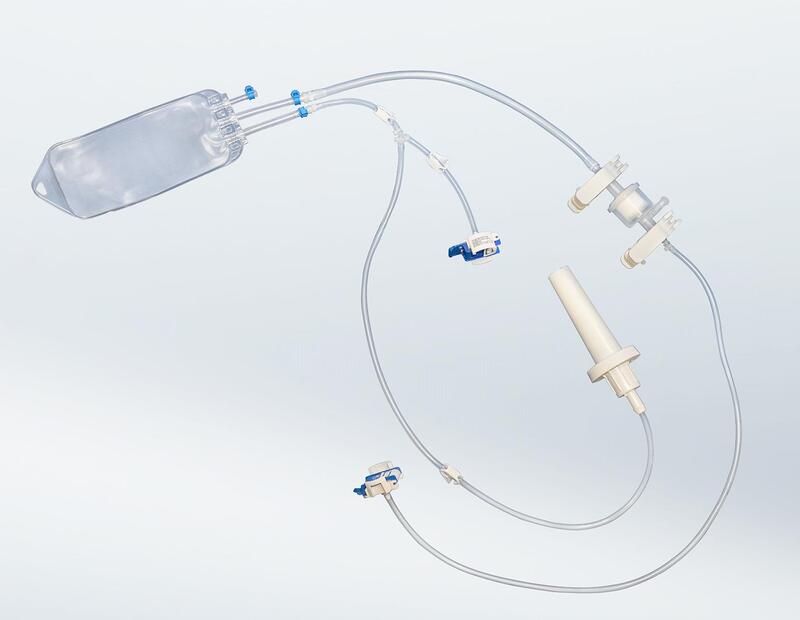
For decades, Aseptic Technologies masters the key requirements for the most critical pharmaceutical ready-to-use devices, their clean production, packaging and sterilization :
We share our excellence with our customers, providing tailored sterilized assemblies in specified packaging :
ISO 9001 : 2015 — Quality management systems
ISO 15378 : 2017 — Primary packaging materials for medicinal products
Read more about Compliance